Sun Jul 26 01:00:06 EDT 2009
testing a battery pack like a man
Blogathon 2009 wordcount: 1397
About a year ago, shortly before I quit my job at lowes, I bought an electric bike kit,[*] since I was, at the time, commuting to work by bicycle, and thought commuting to work by electric bicycle a splendid idea. Once unemployed, my enthusiasm for spending money to commute faster to a job I no longer had held little appeal, and the batteries disappeared into a box in the next move. They remained in that box for the next year, until it occured to me that I really shouldn't have left lead-acid batteries flat for so long, and embarked on a frantic search for them.
*: When I bought it, you could just buy the motor and controller by themselves, but apparently batteryspace has started bundling their battery packs with the motors. I couldn't recommend buying from them before, and I certainly can't now.
Sure enough, when I found them and hooked them up to the charger, it charged for a good five seconds before reporting that the pack was completely charged, which meant, either that the pack was sulfated and now had bugger all capacity, or that the charger was broken.
Fortunately I had a 8 ohm power resistor on hand. Since I=V/R, that means the resistor would draw 4.5 watts at 36 volts, well within the pack's discharge limit, but not so light that it takes forever to run down.
Unfortunately, the power resistor is only rated to 20 watts, and I'll be running 140-odd through it, which would be a problem if resistors went pop and stopped working as soon as you exceeded their power rating. Fortunately, resistors can handle substantially more power with heroic cooling.
Baby, I'm an overclocker.
Heroic cooling is what I do.

The setup is the battery connected to the terminal strip through some Anderson Powerpole connectors, then to the power resistor, with the two 16 gauge wires connecting it to the multimeter.

The resistor itself is suspended in approximately 500 ml of water, which should have enough mass to keep from boiling over the half hour or so it'll take to discharge the pack, not that a mere 100C would damage the resistor.
Following the advice of Derek Lowe, I considered the very worst case scenario of my test setup. That would be a internal short and resultant lead metal fire. Thus, two fire extinguishers,

and a nearby door, for running away.
Thus equipped, I sat down with my trusty pocket-watch, and, for the next twenty-three minutes, wrote down numbers. The results!
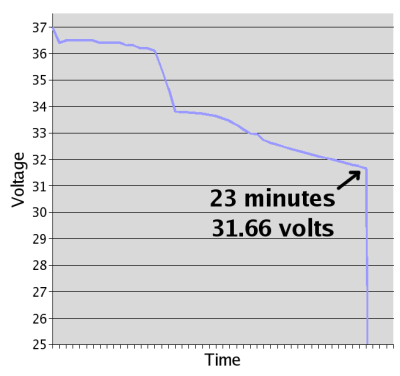
And the raw data [7.4 KiB .ods].
You can tell that it's real science, because the data is so ugly.
Unfortunately, I terminated the test at 10.55 cell voltage, having forgotten that the datasheet graph, at a equivalent discharge, ran the cells down to 9 volts.
This makes it impossible to calculate the real capacity of the battery with any precision, but a rough estimate puts it at 120 amp-hours, well above what would be considered "dead", and pretty close to its original capacity. Science!

The water heat sink got fairly warm (~45-50C) nucleated bubbles on the interior of the glass, and gently steamed, (which I utterly failed to photograph) but otherwise worked perfectly.